INTRODUCTION, EPIDEMIOLOGY, RISK FACTORS
Idiopathic intracranial hypertension (IIH) is a medical condition which manifests with a collection of neurological and ophthalmological symptoms caused by elevated intracranial pressure (ICP) where the cause of the raised ICP is undetectable – thus IIH is a diagnosis of exclusion. IIH is occasionally referred to as pseudotumor cerebri as symptoms often mimic that of brain tumors. IIH almost exclusively occurs in females of childbearing years (20-45) whose body mass index categorizes them as overweight or obese. Median age of diagnosis is 30. Women 10% above ideal weight are thirteen times more likely to develop IIH and this rises to nineteen times more likely for women 20% above their ideal weight. A similar relationship exists amongst men, but to a far lesser degree.
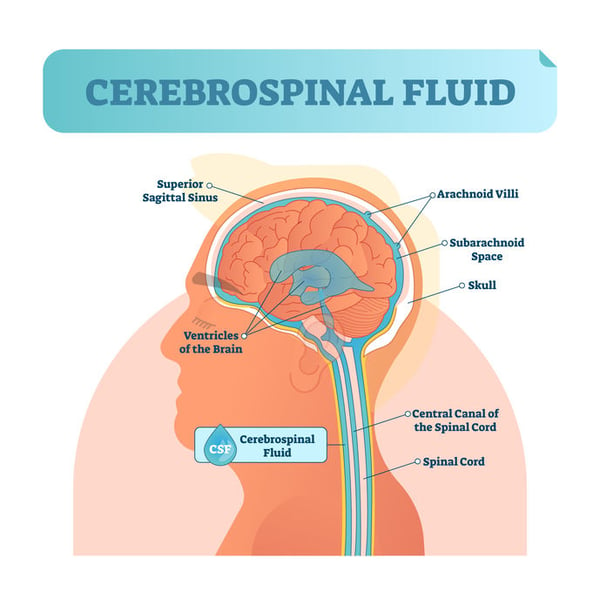
As obesity rates rise worldwide, so has the prevalence of IIH. While the general population prevalence is 1/100,000 persons, the obese female prevalence is 20/100,000. IIH does occur in males and children, but at a substantially lower rate. Only 5% of cases of IIH occur in males. Interestingly the incidence is the same between males and females children.
While the cause of IIH is unknown and is therefore referred to as idiopathic, certain risk factors have been identified, increasing the chance of IIH occurrence. The most notable risk factors include being female, of reproductive age, and being overweight or obese. Recent weight gain between 5-15% of total body weight is linked with IIH. Oral contraceptive pills, tetracycline antibiotics, acne medication, excessive vitamin-A consumption, growth hormone, steroids, lithium, and chemotherapy drugs are associated with IIH. Thyroid dysfunction, chronic kidney failure, dural venous thrombosis, meningitis, lupus, and leukemia are also risk factors. There are no known genetic or ethnicities correlations.
PATHOPHYSIOLOGY OF IIH
The brain and spinal cord are surrounded and cushioned by cerebrospinal fluid (CSF) for protection. CSF is produced in the brain and is eventually absorbed into the bloodstream. IIH occurs when, for no apparent reason, there is excess CSF from increased production, decreased absorption, or impaired venous drainage. Increased ICP may also arise from excessive cerebral blood flow. IIH patients frequently have venous sinuses narrowing; potentially reducing outflow and elevating ICP. Although the underlying mechanism is uncertain, increased fluid in the total cranial space raises ICP and may compress cranial nerves. Moreover, higher ICP causes an abnormal pressure gradient between intraocular pressure and the retrolaminar pressure, thus increasing optic nerve tissue pressure. This interferes with normal axoplasmic transport leading to swelling of the optic disc, called papilledema. Longstanding axoplasmic flow stasis may result in permanent damage in the form of optic atrophy. Papilledema ranges from mild to severe and may result in loss of peripheral, central, and color vision.
DIAGNOSTIC CRITERIA OF IIH
IIH is diagnosed using lumbar puncture, ophthalmoscopy, and neuroimaging.
Lumbar puncture reveals elevated opening CSF pressure, greater than 25 cmH2O. CSF is negative for abnormal cells, indicators of infection, abnormal antibody levels, abnormal glucose concentrations, and abnormal protein levels. Lumbar puncture relieves high ICP and predictably lessens headache symptoms.
Elevated ICP instigates swelling of the optic disc, called papilledema, which is visualized with ophthalmoscopy.
Except for cranial nerve dysfunction, neurological examination is typically unremarkable. Magnetic resonance imaging (MRI) or computed tomography (CT) confirms absence of intracranial masses, but may demonstrate posterior globe flattening, empty sella, perioptic arachnoid space distention, attenuation of cerebral venous sinuses, or optic nerve buckling.
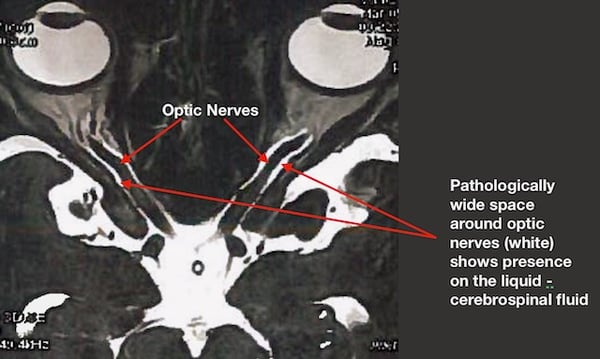
Visual field testing (perimetry) may exhibit visual field defects. 6th cranial nerve involvement may yield horizontal diplopia. Optical coherence tomography (OCT) may visualize swelling and atrophy of the optic nerve.
DIFFERENTIAL DIAGNOSIS FOR IIH
As IIH is a diagnosis of exclusion, underlying causes for elevated ICP must be ruled out. Differential diagnoses include intracranial tumor, intracranial abscess, intracranial hemorrhage, meningitis, choroid plexus papilloma, viral infection, venous drainage obstruction, obstructive hydrocephalus, chronic kidney disease, renal failure, obstructive sleep apnea syndrome, chronic obstructive pulmonary disease, systemic lupus erythematosus, psittacosis, Addison’s disease, adrenal insufficiency, Cushing’s syndrome, hypoparathyroidism, hypothyroidism, hyperthyroidism, Down syndrome, craniosynostosis, and Turner syndrome.
SYMPTOMS OF IDIOPATHIC INTRACRANIAL HYPERTENSION
The most common initial symptom is increasingly painful and frequent headaches not relieved with medication. Headaches occur in over 90% of IIH cases and are usually diffuse and throbbing in nature and often worse in the morning or during activities that raise ICP (sneezing or coughing). Often non-specific, these headaches may mimic other disorders.

Sudden darkening of vision (transient visual obscuration) occurs in 68-72% of cases, unilaterally or bilaterally. 52-87% of patients experience a “whooshing” sensation in one or both ears (pulsatile tinnitus). Other symptoms include blurry vision, dizziness (52%), neck pain (42%), back pain (53%), lightheadedness, nausea, impaired cognition (20%), impaired coordination, decreased color vision, missing peripheral vision, or horizontal diplopia (18%).
CLINICAL SUBTYPES OF IIH
Even if the diagnosis of IIH is made based on exclusion from other medical conditions different clinical types of IIH exist further complicating a final and complete diagnosis and clinical prognosis.
Typical IIH: Female patients of reproductive age with a body mass index greater than 30 kg/m2.
Atypical IIH: Males, females not of childbearing age, and patients of normal body mass index. Meticulous measures are needed to confirm no underlying cause.
Fulminant IIH: Rapid and severe decline of vision within 1 month of diagnosis.
IIH without Papilledema: Very rare. Includes all IIH measures, but without papilledema.
IIH in Ocular Remission: Previously diagnosed IIH with resolved papilledema. Headache symptoms may remain, but vision is no longer threatened.
VISUAL FUNCTION MEASUREMENTS AND MONITORING

Before long-term visual consequences were recognized, IIH was commonly referred to as benign intracranial hypertension. This name was later revised to idiopathic intracranial hypertension as to not overlook the visual complications.
Syndrome of IIH impairs optic nerves causing visual disturbances that should be carefully monitored with specific measures of vision unilaterally.
- Visual acuity: best corrected central vision
- Color vision: ability of differentiating different colors
- Perimetry: peripheral vision sensitivity
While vision is assessed using visual acuity, color vision testing, and perimetry, clinical examination and scans are used to objectively determine severity of IIH. The grade of papilledema is determined with ophthalmoscopy. Fundus photography captures the appearance of the optic nerve, retinal vessels, and macula, to be compared with future pictures to better determine progression or resolution. OCT imaging provides a quick, reliable, accurate, and non-invasive method of measuring the degree of swelling and any loss of ganglion cell or retinal nerve fiber layer (RNFL) thickness. OCT scans are compared at each visit to objectively determine if IIH is progressing or resolving. Ultrasonic B-scanning or CT imaging may be utilized to measure the optic disc height and establish any presence of fluid surrounding the optic nerve.
Reported subjective symptoms of visual disturbances are not specific enough to establish a IIH diagnosis, but when those visual symptoms are combined with mild or severe headaches – one may suspect IIH. Patients may also report transient blurring of central vision only lasting a few seconds and usually early in the morning or following postural changes. Such episodes are called “transient visual obscurations” or “greying out” of vision.
CLASSIFICATION OF VISUAL FIELD ABNORMALITIES
Considerable evidence suggests the principal location of damage from papilledema is the optic nerve head. A conducted study called “Idiopathic Intracranial Hypertension Treatment Trials (IIHTT) verified that structural changes of the optic nerve head with correlated visual field defects are found in IIH. This is further supported by the fact that IIH related visual field defects often occur along the RNFL bundles, similar to glaucoma but with a few exceptions.

IIH related visual field defects range from slight to severe. The majority of these defects are the result of localized nerve fiber layer bundle loss in one hemifield (59.9%). The most frequent visual defect pattern is a partial inferior arcuate defect, and inferior defects occur more frequently than superior defects. Swelling of the optic nerve often produces an enlarged blind spot (15.2%) in the field of vision. Absence of any defects in the field of vision is also possible (11.8%). Sporadic defects may occur, though less often (6.1%). A subset of patients experience diffuse vision loss (5.8%) and few have defects consistent with neurologic-like loss (1.2%). Interestingly, left eyes typically have more visual field loss (62%), postulated to occur due to sleeping position.
Importantly, many visual fields related to IIH defects are reversible if ICP is lowered before optic nerve damage ensues. IIHTT demonstrated that 50% of these visual field defects had reverted back to normal at the one-year mark. However, of the remaining 50%, more than 25% of patients still had visual field defects consistent with RNFL bundle loss.
PAPILLEDEMA
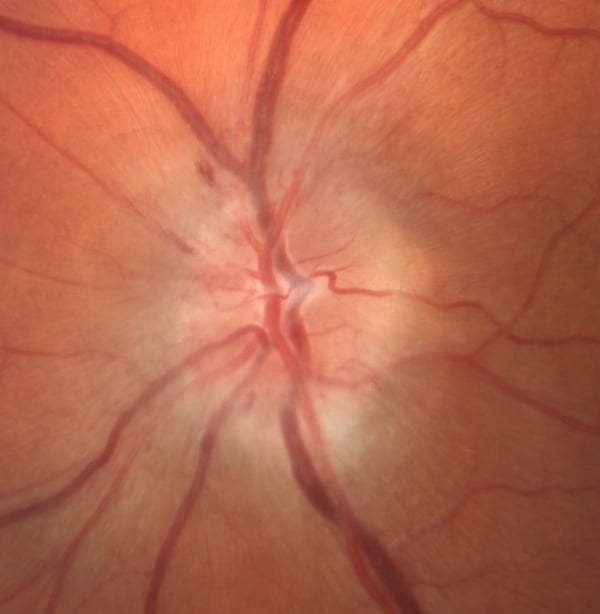
The diagnosis of papilledema specifically refers to the swelling of the optic nerve disc (optic nerve head) due to elevated ICP. Distinguishing papilledema and swelling along the optic nerve is crucial as each has vastly differing implications. Similarly, papilledema must be differentiated from pseudopapilledema, which often occurs from optic disc drusen or a congenital appearance. (IMAGE) A diagnosis of papilledema is concerning as it may be a manifestation of a brain tumor, ischemic damage to the optic nerve from NAION, or inflammation of the central nervous system. As detailed above, it may also be IIH. Neuroimaging is used to reveal the underlying cause or lack of cause (IIH).
Diagnosis: Papilledema is suspected through ophthalmoscopy and confirmed with lumbar puncture measuring high CSF opening pressure. Ophthalmoscopy reveals an elevated optic nerve head with blurred margins and dilated blood vessels. (IMAGE) More severe cases present with obscured retinal vessels, retinal hemorrhages, ischemic areas (cotton wool spots), or hard exudates. If longstanding, disc pallor develops as optic nerve axons atrophy. OCT imaging displays the elevation and abnormally thick RNFL. Visual field defects may be observed with perimetry and color vision testing may be abnormal.
Physical Examination: Blood pressure should be taken in the office to rule out malignant hypertension. Neurological examination is also warranted.
Symptoms: Symptoms of papilledema are similar to those mentioned earlier for IIH, as papilledema occurs with IIH. Few symptoms, if any, present in early papilledema. Headaches occur most frequently and are generally worse in the morning. Other symptoms include blurred vision, transient visual obscurations, horizontal diplopia, pulsatile tinnitus, nausea, difficulty thinking, neck pain, and back pain.
Differential Diagnosis of Papilledema:
- Pseudopapilledema
o Congenital malformation
o Optic nerve drusen
- Malignant hypertension / Hypertensive optic neuropathy
- Optic neuritis
- Papillitis
- Intraocular inflammation (toxoplasmosis, sarcoidosis, uveitis)
- Central retinal vein occlusion
- Ischemic optic neuropathy
- Infiltration of optic disc (lymphoma, leukemia)
- Leber’s hereditary optic neuropathy
- Orbital optic nerve tumors
- Diabetic-related optic neuropathy
- Thyroid-related optic neuropathy
- Optic nerve sheath meningioma
DISTINGUISHING PAPILLEDEMA VS PSEUDOPAPILLEDEMA
While papilledema is optic nerve edema due to elevated ICP, several conditions exist whose optic nerve appearance is similar to papilledema. This finding is largely referred to as pseudopapilledema. Pseudopapilledema is an anomalous optic disc elevation without optic nerve or RNFL edema. Pseudopapilledema typically occurs from a congenital malformation or buried optic disc drusen.(IMAGE)
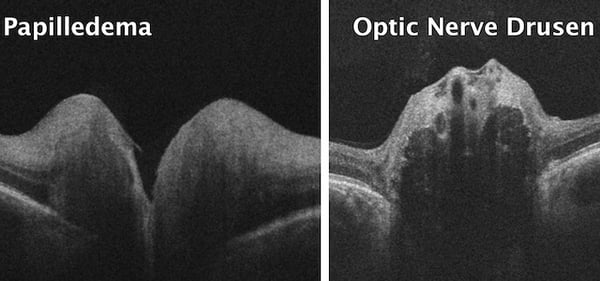
Congenital malformations include a tilted optic disc or hypoplastic optic disc. Optic disc drusen are globular hyaline bodies embedded deep into the optic disc. These, often calcified, drusen frequently give the appearance of the optic disc elevation. Optic disc drusen occur in 2% of the general Caucasian population and are challenging to discern from papilledema. Ultrasound and OCT are both utilized in confirming the presence of optic disc drusen.
Differentiation of papilledema from pseudopapilledema requires clinical expertise and diagnostic testing. Proper diagnosis is crucial as pseudopapilledema is visually unthreatening, while papilledema has serious systemic and visual concerns.
PAPILLEDEMA AND OPTICAL COHERENCE TOMOGRAPHY
Research suggests both qualitative and quantitative measures of peripheral vision may be used to monitor papilledema. Perimetry assesses sensitivity of peripheral vision and is correlated with papilledema grade. OCT measures thickness of the RNFL and overall optic nerve head volume, proven to be exceptionally useful in monitoring and determining papilledema severity. Such highly sensitive and repeatable measurements objectively quantify the degree of progression or resolution. OCT is beneficial for diagnosing papilledema as slight changes in peripapillary retinal pigment epithelium and choroidal folds are observable.
Research supports using both perimetry and OCT for monitoring disc swelling and peripheral vision changes associated with IIH. Such data is also helpful in determining the efficacy of IIH treatments. Moreover, research maintains a correlation between OCT measurements and perimetry results in cases of papilledema.
IDIOPATHIC INTRACRANIAL HYPERTENSION MANAGEMENT
First and foremost, because IIH is a diagnosis of exclusion, other conditions must be ruled out before an IIH diagnosis is made. MRI and CT scans are necessary to rule out a space-occupying lesion.
In overweight or obese patients, the primary goal is sustainable weight loss through dietary and lifestyle modifications. This includes regular exercise, reduced caloric intake, and reduced sodium chloride intake.

Bariatric surgery is used as a last resort, but has been shown to improve symptoms and vision. Treatments also focus on lowering ICP, preserving vision, and relieving headache symptoms. Medical treatment predominantly involves carbonic anhydrase inhibitors such as acetazolamide, which suppresses CSF production up to 57%. IIHTT showed acetazolamide modestly improved vision in mild cases of IIH, but significantly improved vision for moderate to advanced cases of IIH. Importantly, IIHTT determined that acetazolamide plus diet control subjects lost twice the weight than placebo plus diet control subjects. The greatest effect occurred during the first month of treatment and treatment failure was less frequent in subjects taking acetazolamide. Furthermore, subjects not only tolerated the maximum dosages well but their quality of life indicators were significantly better than the control group. The improvement in symptoms outweighed the side effects of acetazolamide. Moreover, dietary changes and acetazolamide significantly improve peripheral vision, optic nerve swelling, and quality of life measures, compared to the control group.
Less commonly, topiramate is used for relieving chronic headaches associated with IIH. Steroid use is generally discouraged, but remains somewhat controversial. Abuse of headache relief medication results in approximately 8% of patients experiencing headaches from medication overuse. Counseling of proper medication usage is needed.
When weight loss and medicinal therapy is not effective or applicable, surgical intervention may be warranted. The most common procedures performed are optic nerve sheath fenestration and neurosurgical shunt implantation.
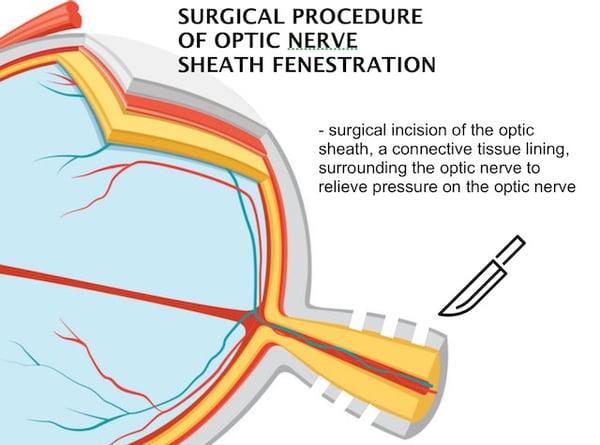
Optic nerve sheath fenestration entails a surgical incision of the optic sheath, a connective tissue lining, surrounding the optic nerve to relieve pressure on the optic nerve. Though highly successful in preserving vision, optic nerve sheath fenestration does not relieve headaches. Neurosurgical shunts may be implanted to drain CSF thus reducing ICP. CSF shunting improves papilledema, RNFL swelling, peripheral vision, and headaches. While often successful in preserving vision and mitigating headaches, a higher complication rate exists. Some evidence shows endovascular lateral venous sinus stenting procedures may protect vision, improve symptoms, and lower ICP in patients with stenosis of venous sinuses by improving the reabsorption of CSF. Repeat stenting is occasionally needed.
Treatment options for IIH are still being investigated. Some researchers have concluded that there is insufficient evidence to strongly recommend or reject any treatment options for IIH at this time.
VISUAL PROGNOSIS
IIH prognosis is highly variable and challenging to predict. Most patients experience improvement in symptoms upon treatment. Some patients experience a continual worsening of symptoms or even resolution and recurrence of symptoms. Approximately 5-10% of female patients encounter disabling visual impairment and 10-25% of patients experience significant long-term vision loss. Untreated or treatment resistant papilledema presents the worse prognosis. Vision loss has historically believed to be permanent, however recent developments suggest otherwise. Most patients never need surgical intervention.
CONCLUSIONS
Since IIH is a diagnosis of exclusion, only upon ruling out other underlying etiologies can a diagnosis be made. IIH poses considerable risk to vision and significant reduction in quality of life when left untreated.

Eye care professionals are often in a unique position to initially encounter these patients as primary symptoms are often related to vision and headaches. When properly treated, headaches may be alleviated, vision may be preserved, and vision loss may even be reversed.
FEDOROV RESTORATION THERAPY
Patients suffering from IIH and unresolved visual disturbances are told no treatment exists for an injured optic nerve and sight can never be recovered or improved. This leaves patients feeling hopeless and without options to restore their vision – but this is far from true. Years of research and practical work has shown that vision loss caused by papilledema due to IIH can be partially reversed resulting in significant improvement in quality of life and visual function. Our clinic in Berlin offers Fedorov Restoration Therapy which is a painless, straightforward therapy designed to improve the function of retinal cells and the entire optic nerve. Treatment involves application of weak electrical current pulses targeting partially-damaged retinal cells to improve the conductivity of signals to the brain thus enhancing visual signals through the damaged optic nerve. The Fedorov Restoration Therapy interdisciplinary approach bridges ophthalmology and neurology, eye and brain, combining the two in order to achieve spectacular results for visually impaired patients afflicted by optic nerve disorders. This is an innovative and completely non-invasive, non-surgical method that improves and restores vision naturally with stable results and with no risk of side effects. If you or a loved one are afflicted by visual impairment, especially from optic nerve damage, Fedorov Restoration Therapy might be the perfect treatment for you.
Blog prepared in cooperation with Kaleb Abbott, O.D., M.S.
REFERENCES
- Wall M. Idiopathic intracranial hypertension and the idiopathic intracranial hypertension treatment trial. J Neuroophthalmol. 2013;33(1):1-3.
- Hung HL, Kao LY, Huang CC. Ophthalmic features of idiopathic intracranial hypertension. Eye (Lond). 2003;17(6):793-5.
- Keltner JL, Johnson CA, Cello KE, Wall M. Baseline visual field findings in the Idiopathic Intracranial Hypertension Treatment Trial (IIHTT). Invest Ophthalmol Vis Sci. 2014;55(5):3200-7.
- Wall M. Update on Idiopathic Intracranial Hypertension. Neurol Clin. 2017;35(1):45-57.
- Wall M, Johnson CA, Cello KE, et al. Visual Field Outcomes for the Idiopathic Intracranial Hypertension Treatment Trial (IIHTT). Invest Ophthalmol Vis Sci. 2016;57(3):805-12.
- Cello KE, Keltner JL, Johnson CA, Wall M. Factors Affecting Visual Field Outcomes in the Idiopathic Intracranial Hypertension Treatment Trial. J Neuroophthalmol. 2016;36(1):6-12.
- Mollan SP, Davies B, Silver NC, et al. Idiopathic intracranial hypertension: consensus guidelines on management. J Neurol Neurosurg Psychiatry. 2018;89(10):1088-1100.
- Hung HL, Kao LY, Huang CC. Ophthalmic features of idiopathic intracranial hypertension. Eye (Lond). 2003;17(6):793-5.
- Mollan SP, Markey KA, Benzimra JD, et al. A practical approach to, diagnosis, assessment and management of idiopathic intracranial hypertension. Pract Neurol. 2014;14(6):380-90.
- Rebolleda G, Muñoz-negrete FJ. Follow-up of mild papilledema in idiopathic intracranial hypertension with optical coherence tomography. Invest Ophthalmol Vis Sci. 2009;50(11):5197-200.
- Friedman DI, Mcdermott MP, Kieburtz K, et al. The idiopathic intracranial hypertension treatment trial: design considerations and methods. J Neuroophthalmol. 2014;34(2):107-17.




1 Comment
Click here to read/write comments